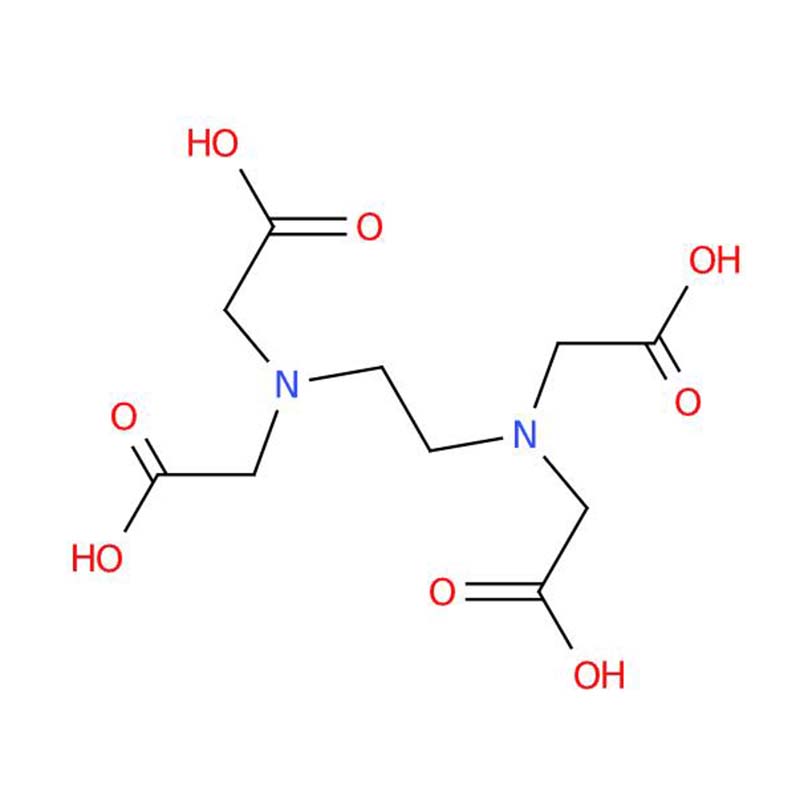
Ethylenediaminetetraacetic acid CAS:60-00-4
Potent Chelating Capacity: EDTA possesses hexadentate chelating properties, forming highly stable coordination complexes via six-point binding interactions with metal ions.
Efficient Ligand Function: Acting as a powerful polydentate ligand, it is extensively applied in industrial and laboratory environments for the complexation of metal ions.
Broad Applicability: This compound is utilized across multiple diverse fields, including water treatment processes, pharmaceutical production, agricultural formulation development, and as a specialized additive in chemical reactions.
Robust Chemical Stability: EDTA retains its structural intactness at elevated temperatures (up to 250°C) and shows resistance to degradation, ensuring consistent performance in rigorous application scenarios.
Ethylenediaminetetraacetic Acid (EDTA) is known as a very efficient polydentate ligand in aqueous solution. When deprotonated, it becomes the EDTA⁴⁻ anion, featuring six potential binding sites—four of which are provided by carboxylate oxygen atoms, and two by amine nitrogen atoms—that can interact with metal ions simultaneously.
Ethylenediaminetetraacetic acid Chemical Properties
Melting point | 250 °C (dec.) (lit.) |
Boiling point | 434.18°C (rough estimate) |
bulk density | 600kg/m3 |
density | 1.46 g/cm3 at 20 °C |
vapor pressure | <0.013 hPa (20 °C) |
refractive index | n20/D 1.363 |
Fp | >400°C DIN 51758 |
storage temp. | 2-8°C |
solubility | 3 M NaOH: 100 mg/mL |
form | crystalline |
pka | pKa 2 (Uncertain);10.26 (Uncertain) |
color | White to almost white |
PH | 2.5 (10g/l, H2O, 23℃)(slurry) |
Odor | Odorless |
PH Range | 2.5 at 10 g/l at 23 °C |
Water Solubility | 0.5 g/L (25 ºC) |
λmax | λ: 280 nm Amax: ≤0.25 |
Decomposition | 240 °C |
Merck | 14,3517 |
BRN | 1716295 |
Stability: | Stable. Incompatible with copper, copper alloys, nickel, aluminium, strong oxidizing agents, strong bases |
LogP | -0.836 (est) |
CAS DataBase Reference | 60-00-4(CAS DataBase Reference) |
NIST Chemistry Reference | N,N'-1,2-Ethane diylbis-(N-(carboxymethyl)glycine)(60-00-4) |
EPA Substance Registry System | Ethylenediaminetetraacetic acid (60-00-4) |
Safety Information |
Hazard Codes | Xi |
Risk Statements | 36-52/53-36/37/38-36/38 |
Safety Statements | 26-61-37/39-36 |
RIDADR | UN 3077 9 / PGIII |
WGK Germany | 2 |
RTECS | AH4025000 |
F | 3 |
Autoignition Temperature | >200 °C |
TSCA | Yes |
HS Code | 2922 49 85 |
Hazardous Substances Data | 60-00-4(Hazardous Substances Data) |
Toxicity | LD50 orally in Rabbit: 2580 mg/kg |
In chelation, the EDTA anion employs all six coordination sites to encapsulate a metal ion, generating remarkably stable complexes. This multidentate mode yields a claw‑like grip around the metal center, analogous to a crustacean's pincer—the source of the term "chelation." The high stability of EDTA‑metal complexes follows directly from this maximized coordination, as it allows six‑point binding to the target ion.