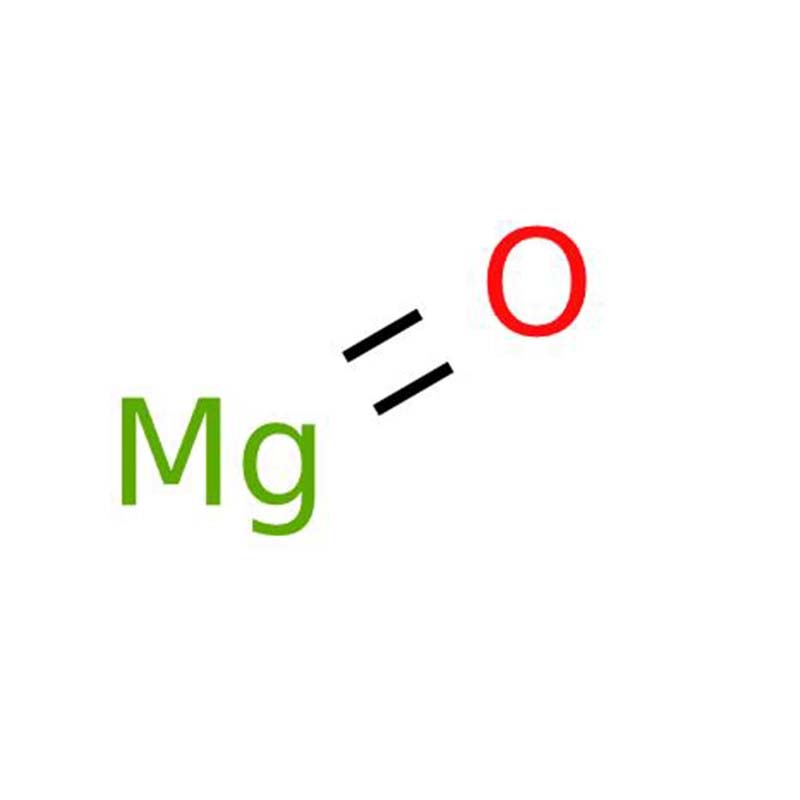
Magnesium oxideCAS:1309-48-4
Material Performance and Application Introduction
Outstanding High-Temperature Resistance
This material boasts an extremely high melting point reaching 2,852°C. Thanks to its superior thermal stability at extreme temperatures, it is perfectly applicable to the production of refractory materials and high-temperature crucibles, meeting the strict usage demands of high-temperature industrial scenarios.
Multi-Purpose Industrial Functional Ingredient
It can be added to rubber and plastic composite materials to significantly optimize their comprehensive performance. In the manufacturing process of silicon steel products, the material also acts as an efficient catalyst and insulating medium, playing dual functional roles in industrial production and processing.
Excellent Fire-Retardant Capability
When blended into various building raw materials, it can effectively upgrade the fireproof performance of finished construction products, greatly enhancing the fire safety level of building structures and complying with industrial fire protection standards.
Wide Application in Medical and Agricultural Fields
In the pharmaceutical industry, it can be prepared into medicinal formulations as an effective antacid ingredient. In agriculture, it is widely used for soil improvement and livestock and poultry feeding, serving as a high-quality magnesium source supplement to optimize soil quality and promote animal growth.
Diversified Customized Specifications
Two mainstream product types are provided for differentiated industrial needs: light-grade products with larger specific surface area and heavy-grade products featuring higher bulk density. The graded design enables targeted matching with diverse professional application scenarios and technical requirements.
Magnesium oxide, also widely known as magnesia, is an essential oxide of alkaline earth metals with the chemical formula MgO. Appearing as a white crystalline powder, this substance features outstanding thermal resistance, with a melting point as high as 2,852°C and a boiling point of 3,600°C, alongside a specific gravity of 3.58 under standard room temperature conditions of 25°C.
In terms of chemical solubility, the compound can dissolve in acid solutions and ammonium salt liquids. When exposed to water, it will slowly hydrate and convert into magnesium hydroxide. Furthermore, magnesium oxide is capable of producing magnesium bicarbonate after reacting with water saturated with carbon dioxide.
This chemical substance is hygroscopic in nature. It can continuously absorb moisture and carbon dioxide from the ambient air during storage and application. In addition, thermal decomposition of magnesium oxide will release irritating gaseous fumes, which is a notable property requiring attention in practical operation and high-temperature processing.
Magnesium oxide is mainly manufactured from raw materials including magnesite (MgCO₃), dolomite (MgCO₃·CaCO₃) and seawater resources. In industrial manufacturing, the mainstream technique is the high-temperature calcination of magnesite or dolomite ores to trigger thermal decomposition reactions for product preparation. Another mature preparation approach takes seawater as the raw material: calcium hydroxide is added to seawater to precipitate magnesium hydroxide intermediates, which are then calcinated to obtain finished magnesium oxide products. Moreover, magnesium chloride brines recycled from seawater or bromine extraction waste liquor can also be adopted for production. By adding sodium hydroxide or sodium carbonate to such brines, magnesium hydroxide and basic magnesium carbonate precipitates can be generated separately, and these precipitates can be further transformed into magnesium oxide via high-temperature heating treatment. At present, domestic manufacturers in China primarily adopt magnesite, dolomite, natural brine and solid salt ore as core raw materials for magnesium oxide production.
As one of the most vital magnesium-containing chemical products, magnesium oxide ranks first in production volume, occupying roughly 75% of the total output of industrial magnesium chemicals. Products calcinated at temperatures lower than 900 °C are defined as light-burned magnesia. This material features low bulk density, large specific surface area and excellent adsorption performance, endowing it with versatile industrial applicability. It can be used as an efficient catalyst and reinforcing filler for rubber composite materials, and serves as a core raw material for preparing magnesium oxychloride cement when mixed with magnesium chloride solution. Beyond industrial utilization, light-burned magnesia is widely applied in construction, medical and agricultural fields. It can be blended into building materials to achieve flame retardant effects, and applied in pharmaceutical preparations to relieve gastric hyperacidity and treat peptic ulcer diseases. In most medical formulas, it is compounded with calcium carbonate to alleviate the laxative side effect of pure magnesium oxide. In agriculture, it is commonly added to livestock feed and crop fertilizers to supplement essential magnesium nutrients for animals and plants.
Magnesia calcined at 950–1050 °C is classified as light-burned magnesia. This material possesses a higher bulk density, stable particle size distribution, and improved hydration activity. In industrial practice, it is widely utilized as a release agent for silicon steel sheets. During high-temperature treatment, it can react with silicon dioxide on the steel surface to generate a magnesium silicate protective film. The formed barrier layer effectively avoids adhesion and sintering of silicon steel substrates under extreme heating conditions, ensuring the processing quality of finished products.
In contrast, heavy-burned magnesia is synthesized under high calcination temperatures ranging from 1500 °C to 1800 °C. Benefiting from ultra-high-temperature sintering, this material features a large volume density and a relatively small specific surface area. It delivers outstanding thermal stability and low chemical activity, which is reflected in its slow hydration rate and low solubility in acidic solutions. Owing to these superior physical and chemical properties, heavy-burned magnesia is an ideal refractory raw material, which is extensively applied in the manufacturing of high-temperature crucibles and industrial furnace lining materials for high-temperature industrial scenarios.